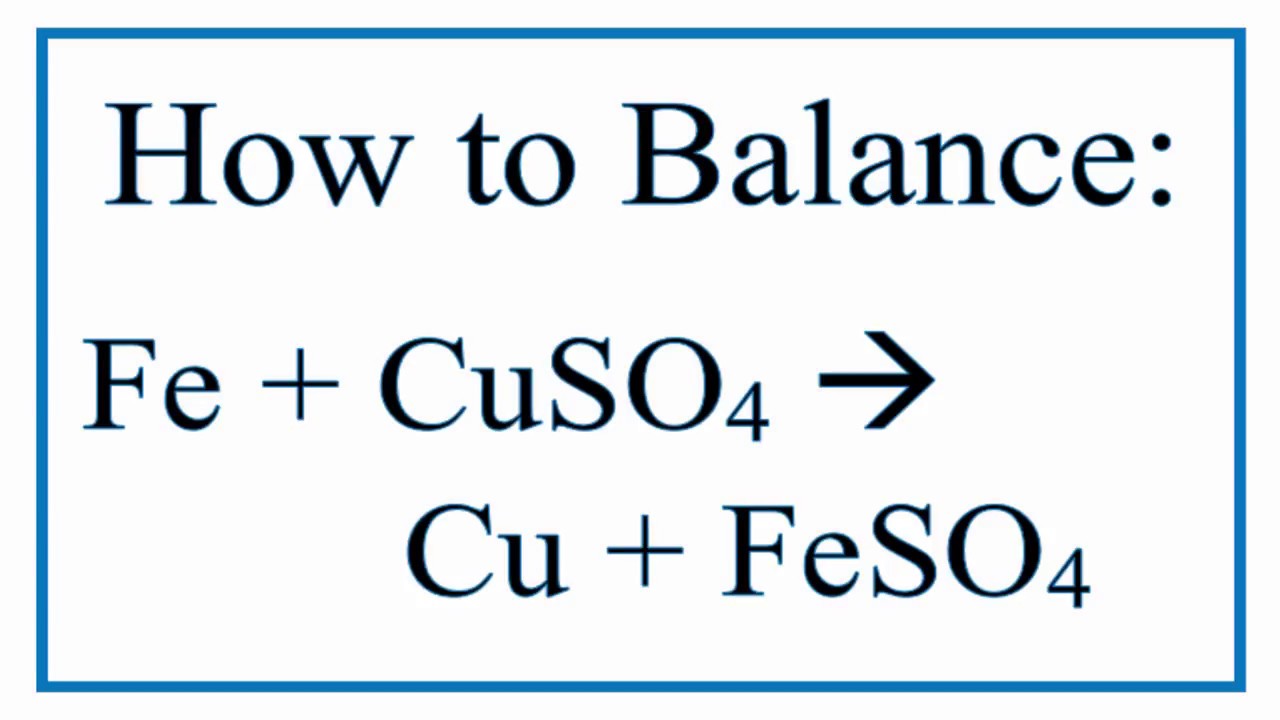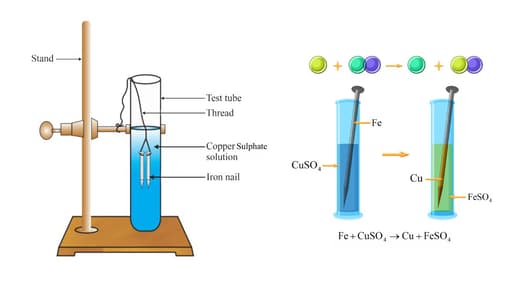
Isha and Rajivwere experimenting in the laboratory They decided to do two different experiments to find out the reactivity of the metals Ishaplaced iron nailsin a copper sulphate solution Rajivplacedcopper nails in

Write a neat diagram and explain how the change in colour of the copper sulphate solution due to reaction with iron.
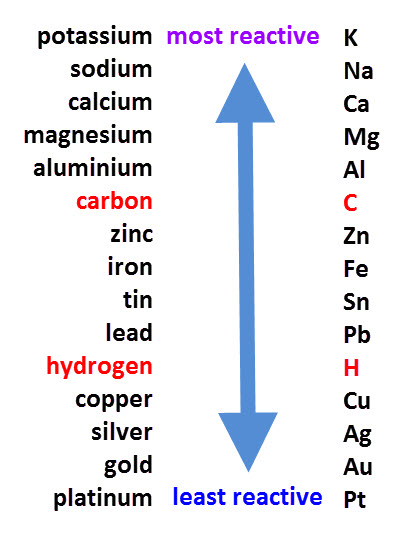
Chemical reaction takes place when iron dust is added into copper sulphate solution but no reaction takes place when copper dust is added into ferrous sulphate solution, why? | Socratic

Purpose – To react copper sulfate with iron, producing iron sulfate and copper. Materials – copper sulfate (CuSO 4 ) and Fe filings – water – beakers, - ppt download
What changes in colour of iron nails and copper sulphate solutions do you observe after keeping the iron nails dipped in copper sulphate solution for about 30 minutes? - Quora

Class 9 Chemistry Practical's and Experiments on To carry out the reaction of Iron with copper sulphate solution in water and classify it as physical or chemical changes